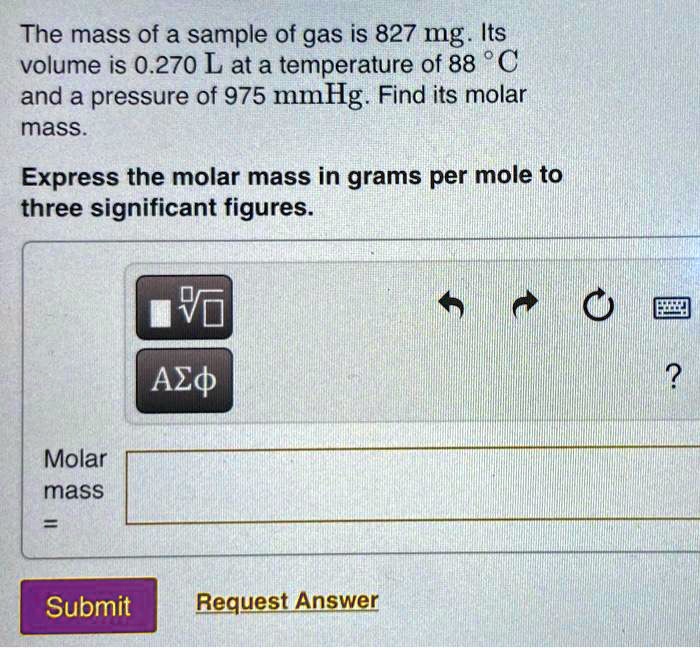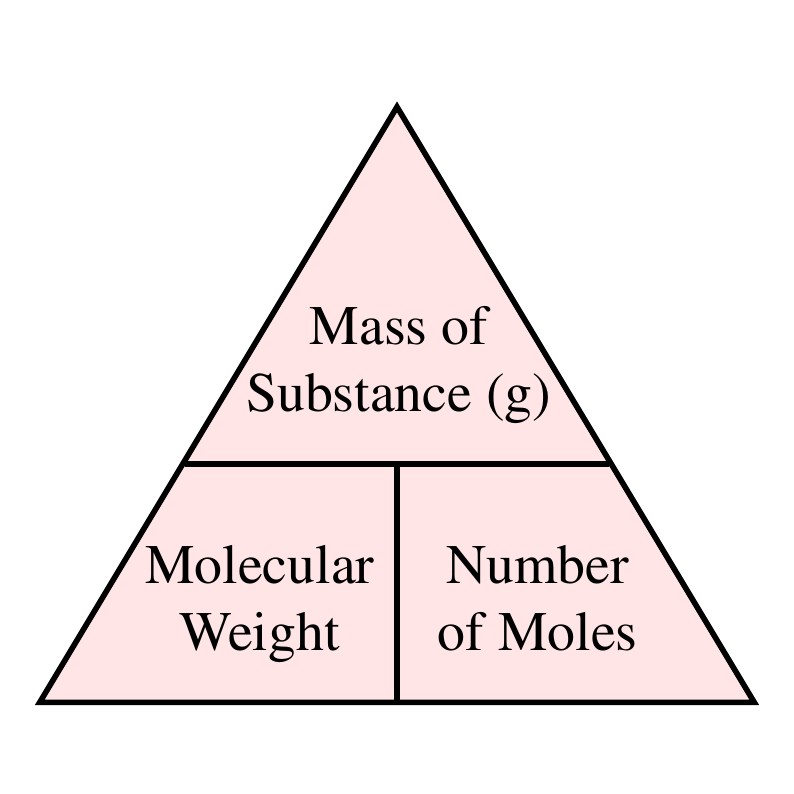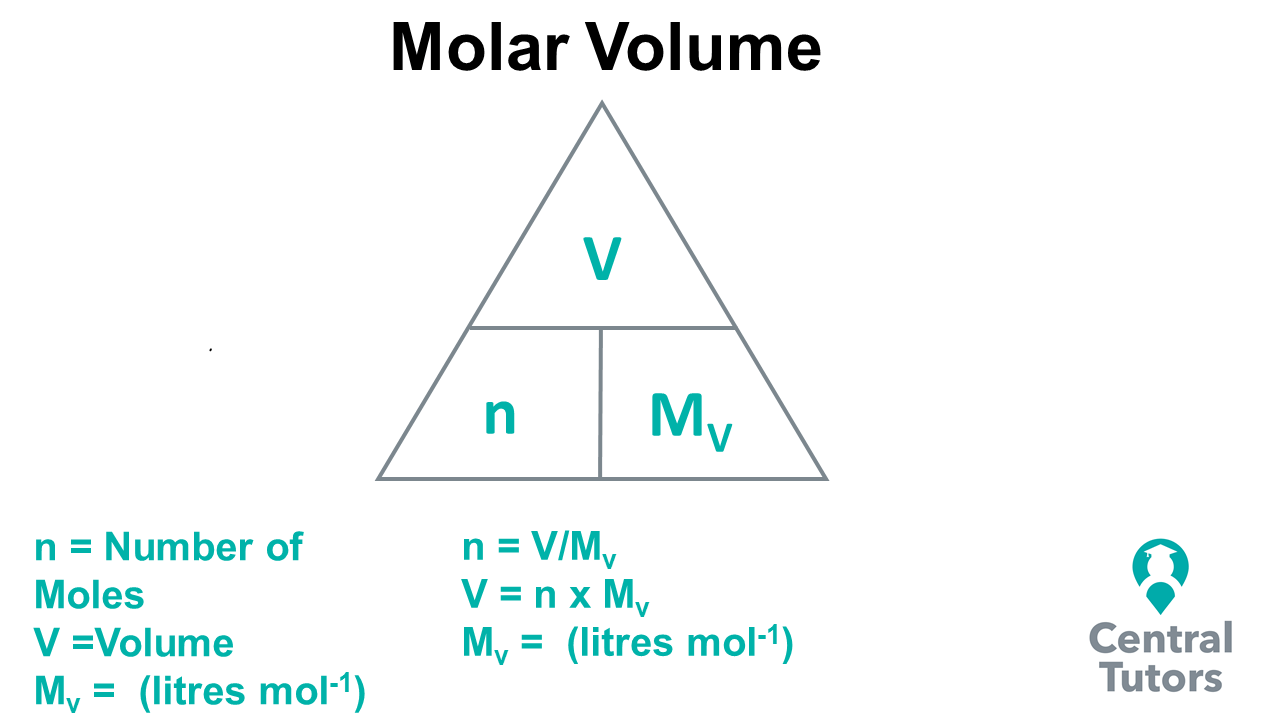
SOLVED: Review Consan One mole of any gas has volume of 22.4 Lat STP What are the molar masses of the following gases, and what are their densities in grams per liter

Molarity Dilution Problems Solution Stoichiometry Grams, Moles, Liters Volume Calculations Chemistry - YouTube

Review: Mole Conversions: Convert 3 mols Oxygen to grams: Convert 42 grams Chlorine to mols: What is % composition? What is the %comp of magnesium in magnesium. - ppt video online download