All-oral longer regimens are effective for the management of multidrug-resistant tuberculosis in high-burden settings | European Respiratory Society
WHO's Rapid Communication on Treatment of Multidrug- and Rifampicin-Resistant Tuberculosis | Stop TB Partnership

Frontiers | Comparative effectiveness of individualized longer and standardized shorter regimens in the treatment of multidrug resistant tuberculosis in a high burden country

Annex. Summary of changes in policy on DS-TB treatment since 2010 and mapping of recommendations in consolidated DS-TB guidelines | TB Knowledge Sharing

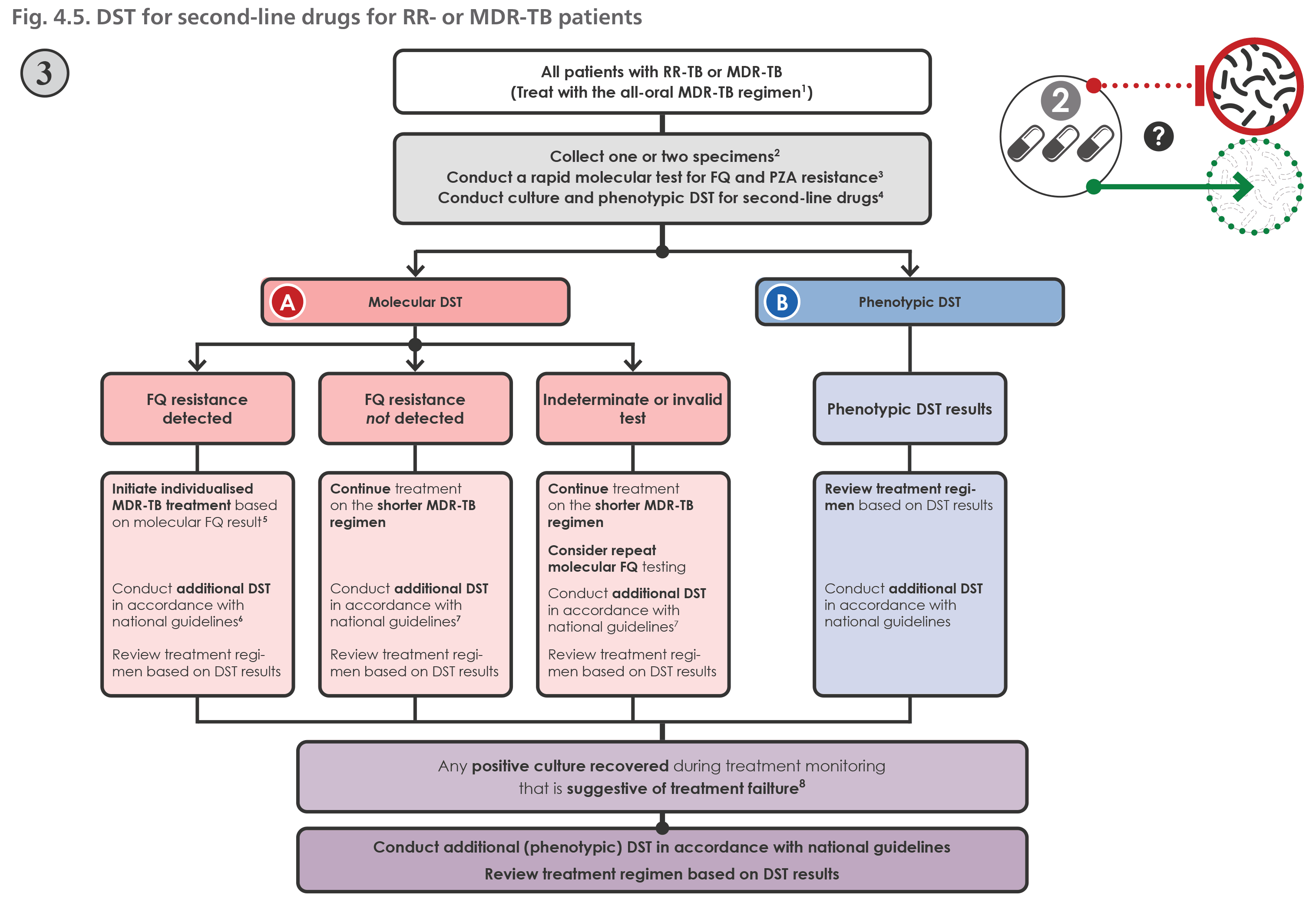
Fig. 4.1, Criteria to decide when the shorter MDR-TB regimen may be offered - WHO consolidated guidelines on drug-resistant tuberculosis treatment - NCBI Bookshelf
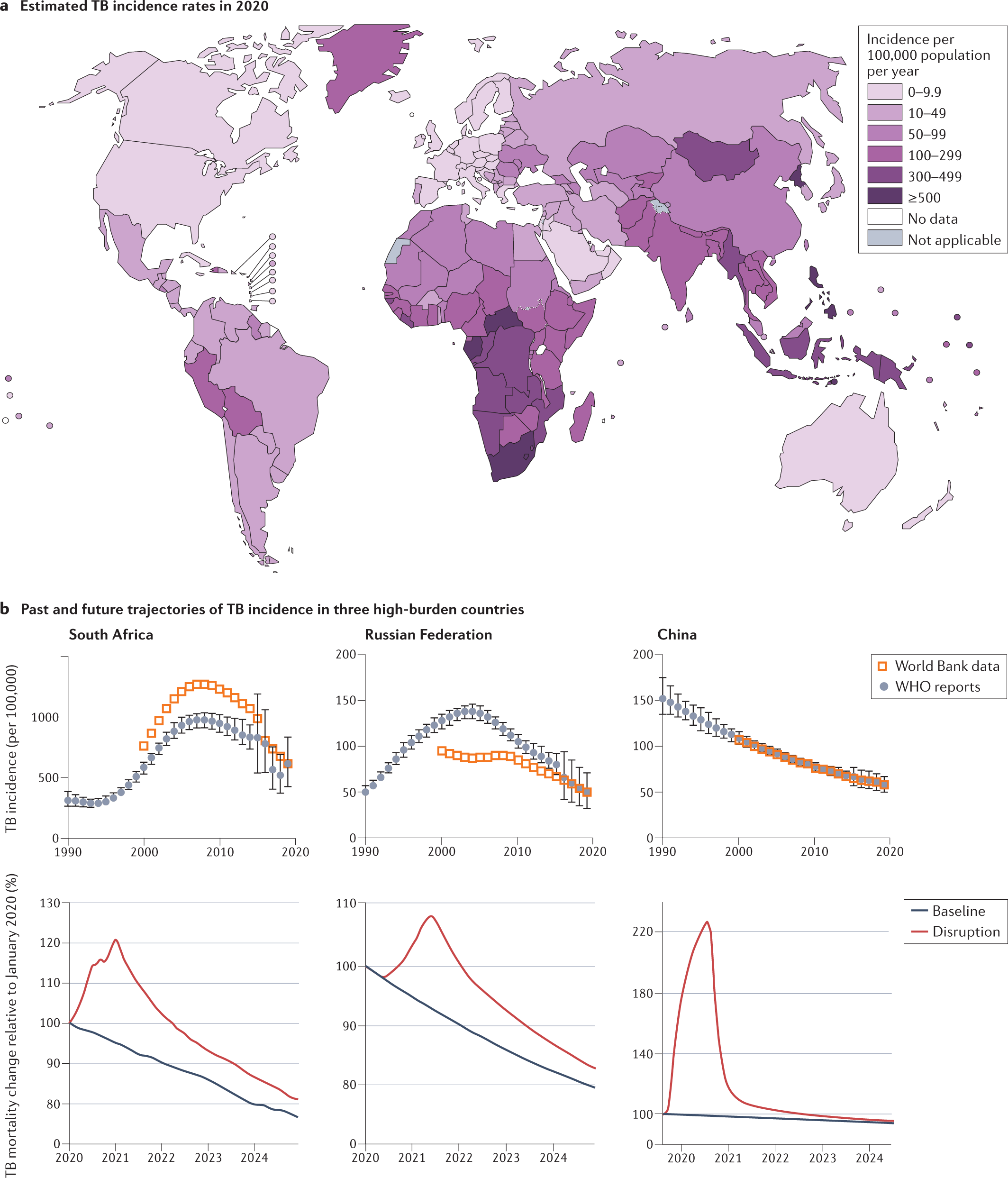
Anti-tuberculosis treatment strategies and drug development: challenges and priorities | Nature Reviews Microbiology

Drug-associated adverse events in the treatment of multidrug-resistant tuberculosis: an individual patient data meta-analysis - The Lancet Respiratory Medicine

MDR/XDR-TB management of patients and contacts: Challenges facing the new decade. The 2020 clinical update by the Global Tuberculosis Network - ScienceDirect

Keeping up with the guidelines: design changes to the STREAM stage 2 randomised controlled non-inferiority trial for rifampicin-resistant tuberculosis | Trials | Full Text
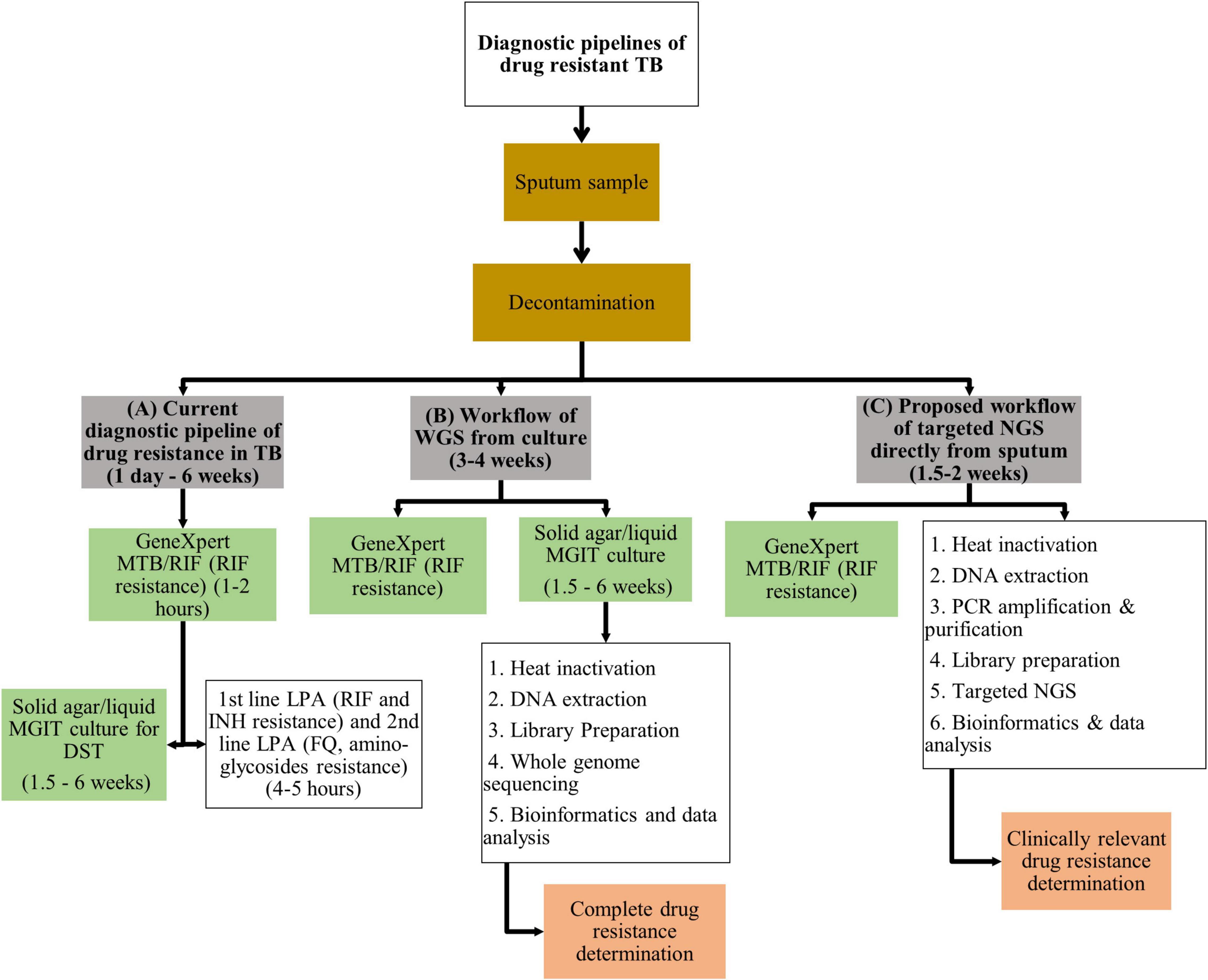
Frontiers | Application of Next Generation Sequencing for Diagnosis and Clinical Management of Drug-Resistant Tuberculosis: Updates on Recent Developments in the Field

Timeline of changes to trial design and release of WHO treatment guidelines | Download Scientific Diagram

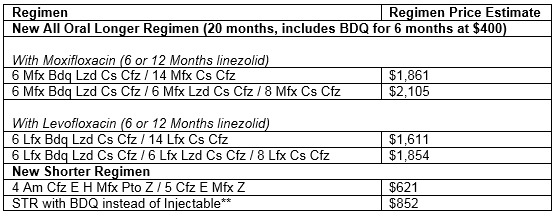
Drugs and doses used in shorter MDR regimen for patients older than 14... | Download Scientific Diagram

9 months of delamanid, linezolid, levofloxacin, and pyrazinamide versus conventional therapy for treatment of fluoroquinolone-sensitive multidrug-resistant tuberculosis (MDR-END): a multicentre, randomised, open-label phase 2/3 non-inferiority trial in ...
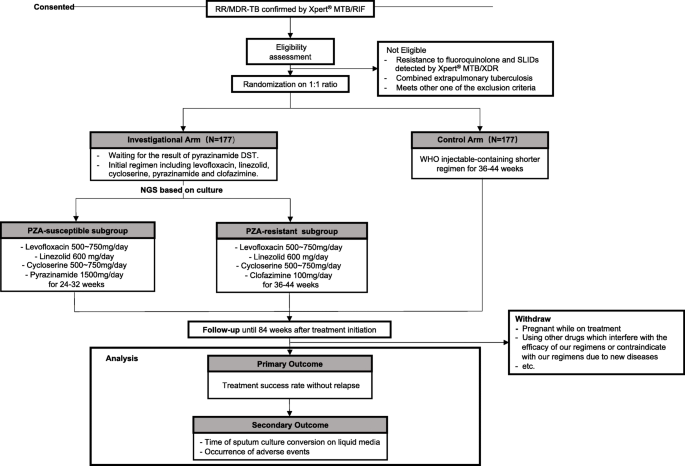
Refining MDR-TB treatment regimens for ultra short therapy (TB-TRUST): study protocol for a randomized controlled trial | BMC Infectious Diseases | Full Text