Li Lithium Chemical Element Periodic Table. Single vector illustration, colorful Icon with molar mass, electron conf. and atomic number Stock Vector Image & Art - Alamy

SOLVED:The atomic mass of lithium on a four-significant-figure periodic table is 6.941 amu. Lithium has two natural isotopes with atomic masses of 6.10512 amu and 7.01600 amu. Calculate the percentage distribution between
Lithium exists as two isotopes: Lithium-6 and Lithium-7 in the ratio of 2:25. What is the relative atomic mass of the lithium? - Quora










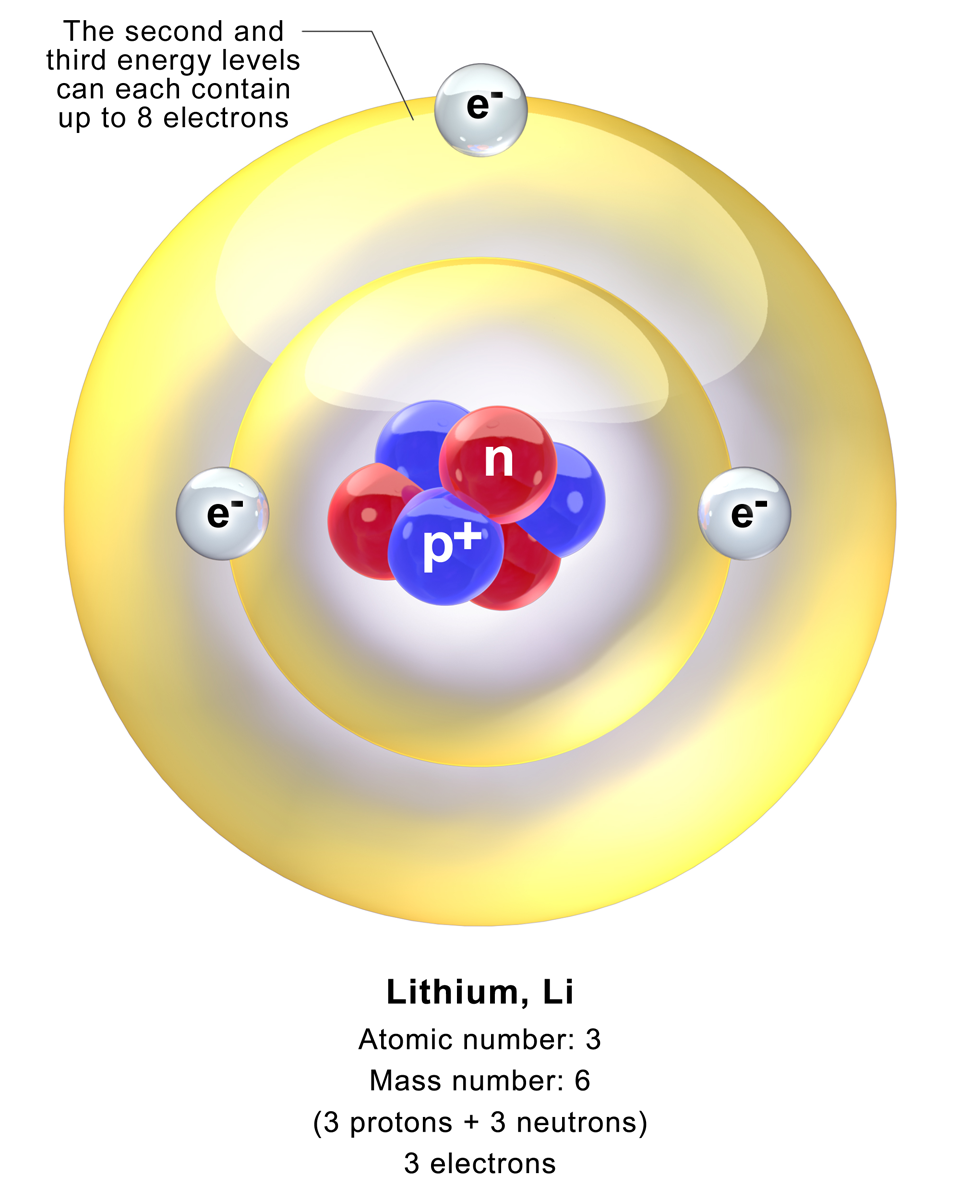





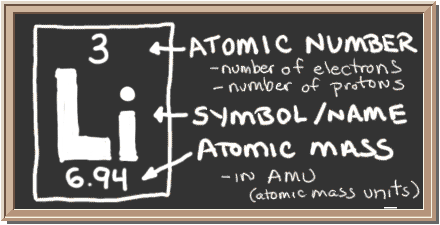

/lithium--illustration-545864483-570a6bde3df78c7d9edbafc1.jpg)
