PDF) Updating Framingham CVD risk score using waist circumference and estimated cardiopulmonary function: a cohort study based on a southern Xinjiang population

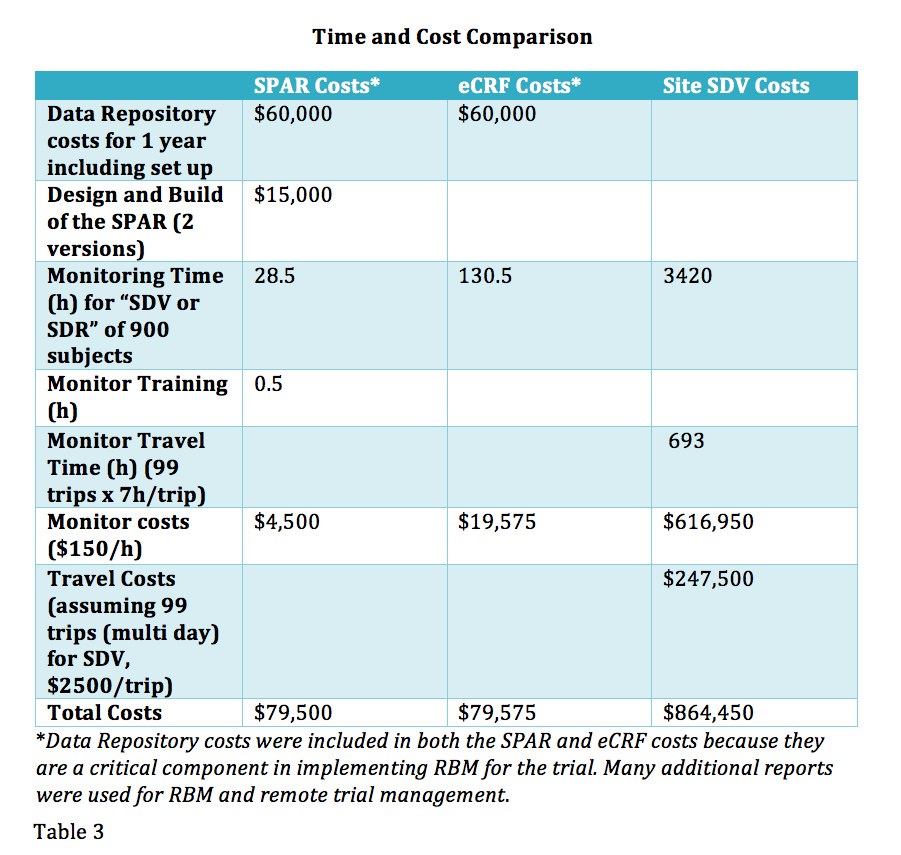
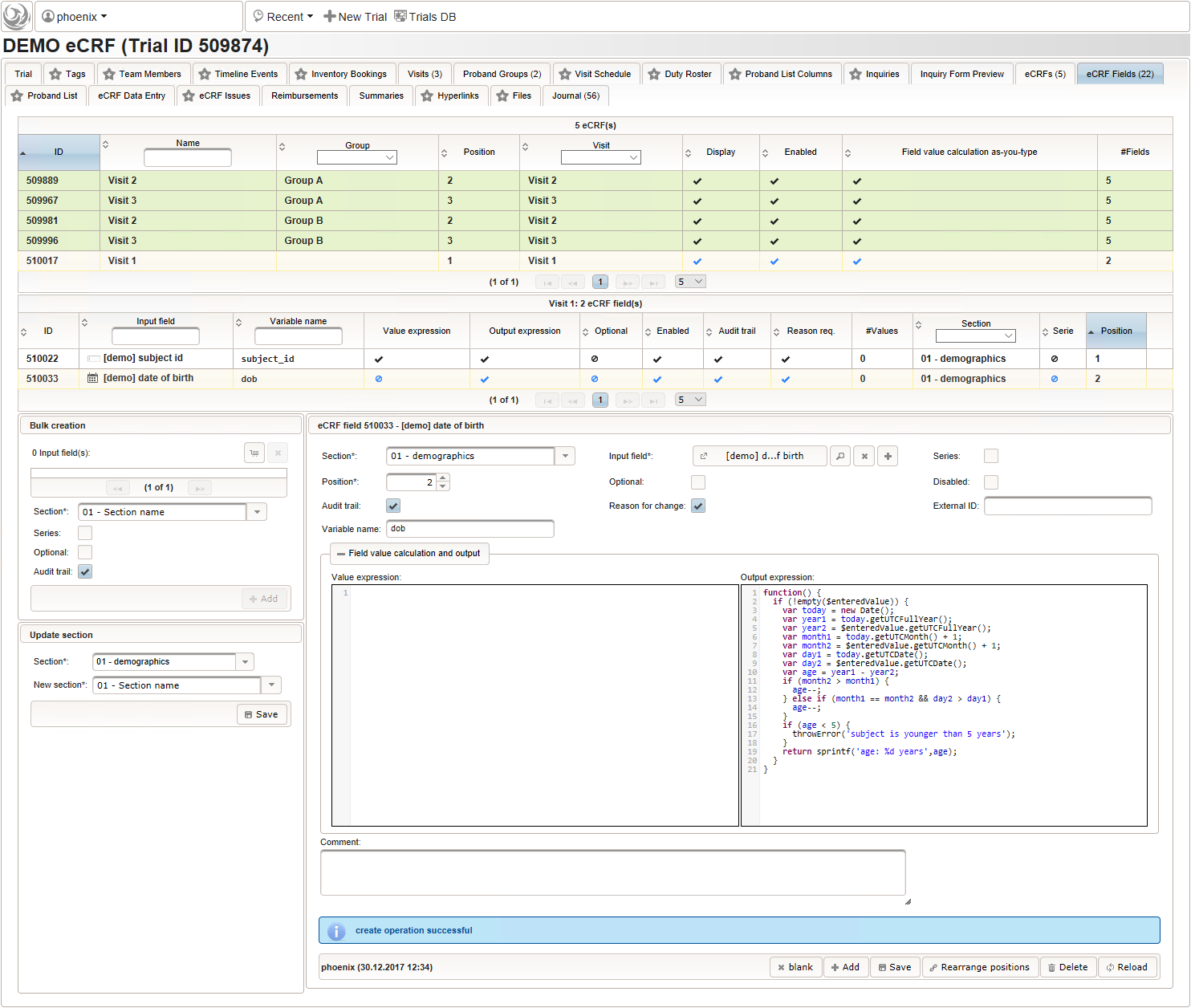
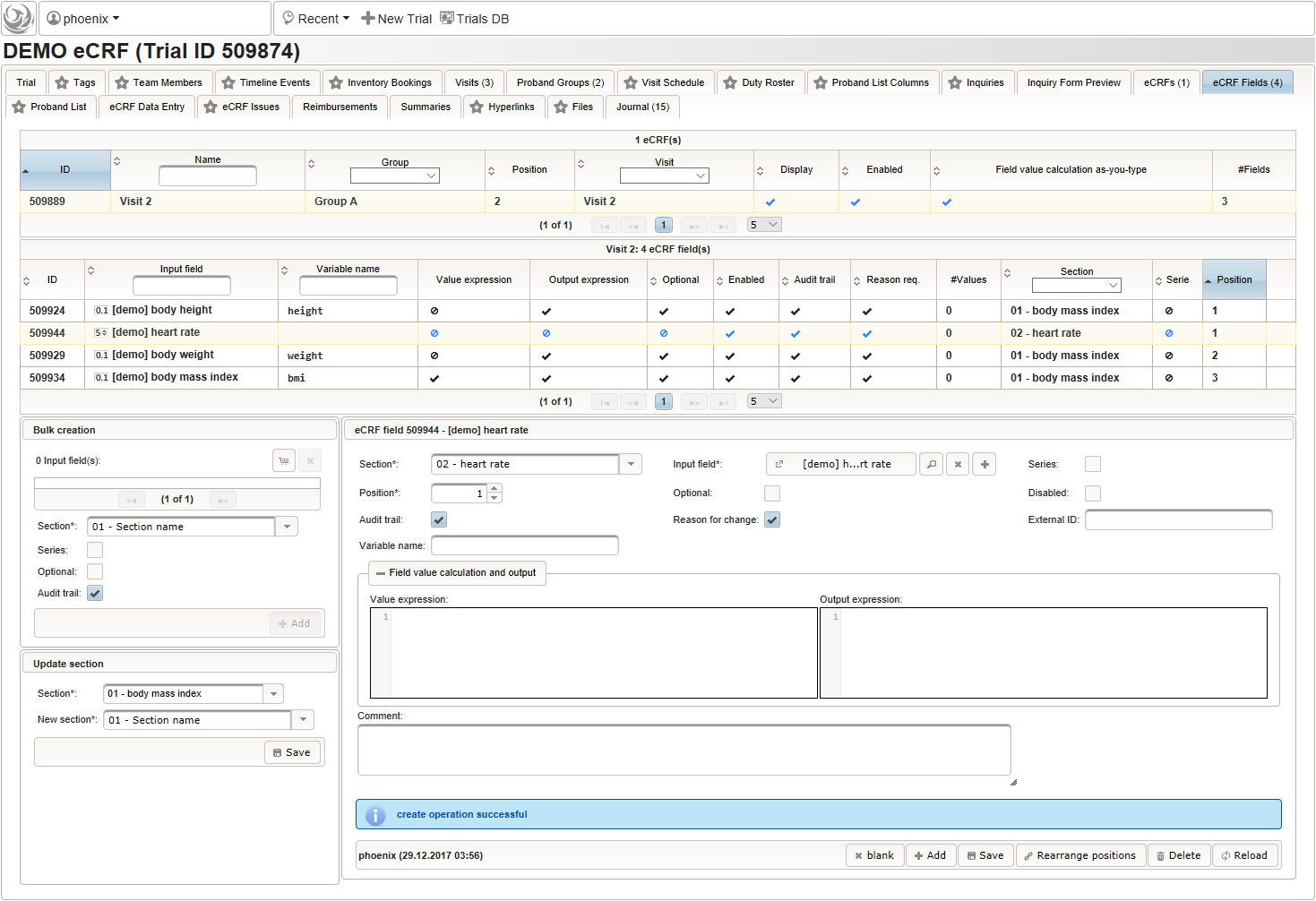
A multi-technique approach to bridge electronic case report form design and data standard adoption - ScienceDirect

A multi-technique approach to bridge electronic case report form design and data standard adoption - ScienceDirect