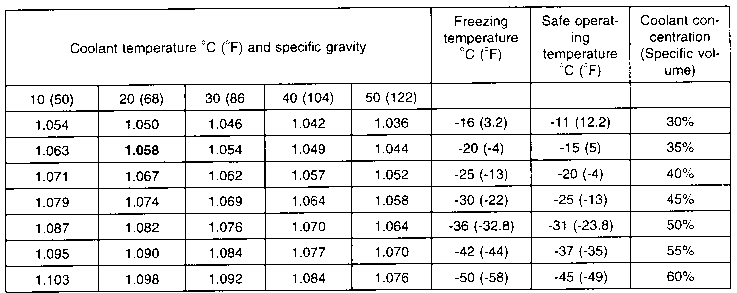
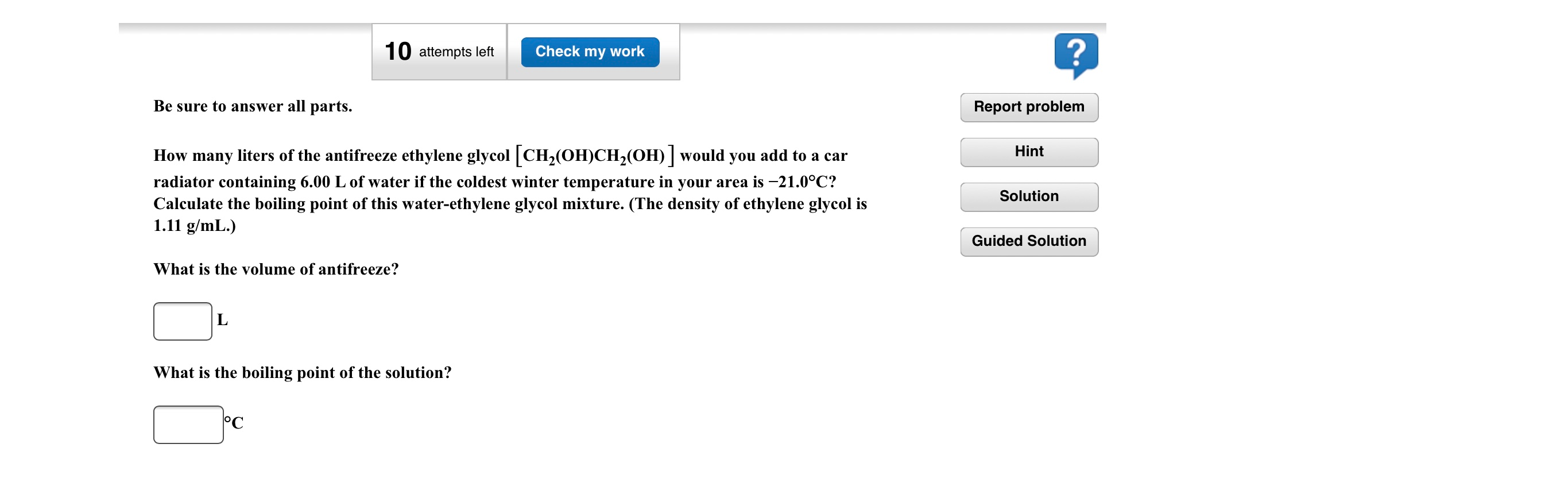
![SOLVED:How many liters of the antifreeze ethylene glycol [CH2(OH) CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is -20^∘ SOLVED:How many liters of the antifreeze ethylene glycol [CH2(OH) CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is -20^∘](https://cdn.numerade.com/previews/f6f43457-93cd-4816-bbe1-0388c635c862_large.jpg)
SOLVED:How many liters of the antifreeze ethylene glycol [CH2(OH) CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is -20^∘

OneClass: General Chemistry II Homework 1. You add 500. g of ethylene glycol, C H,O,(i.e. antifreeze)...

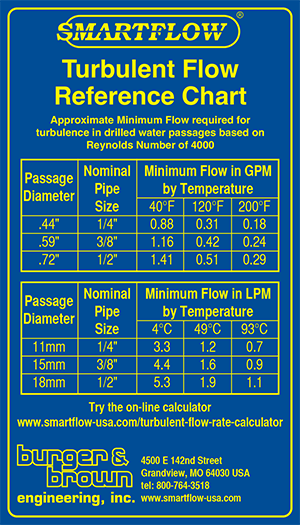
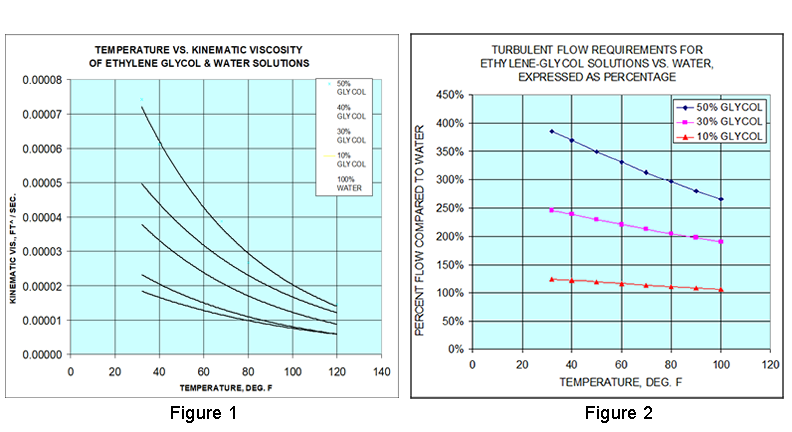
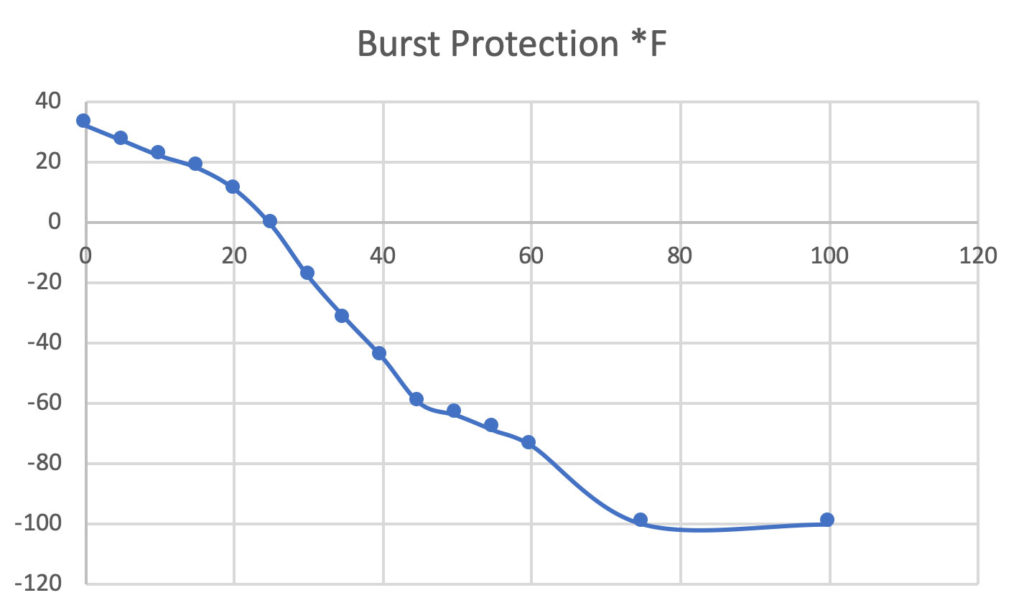
Antifreeze Refractometer Ethylene Glycol: -84°F-32°F 0-70% VOL ,Propylene Glycol: -60°F-32°F 0-63% VOL with ATC, they can be used to test the concentration of ethylene glycol or propylene glycol in antifreeze liquids. :







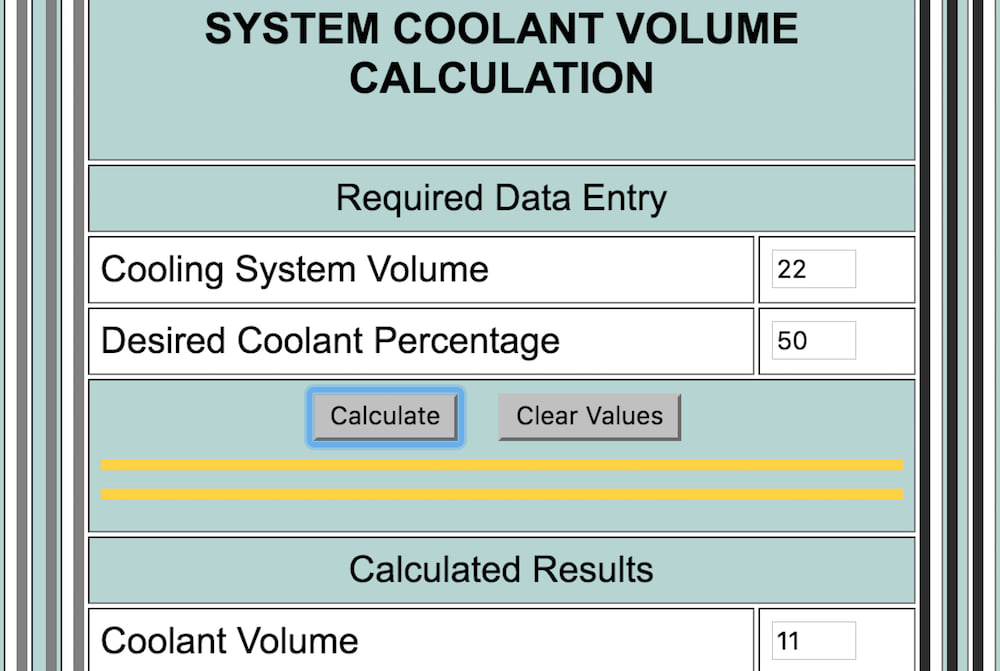

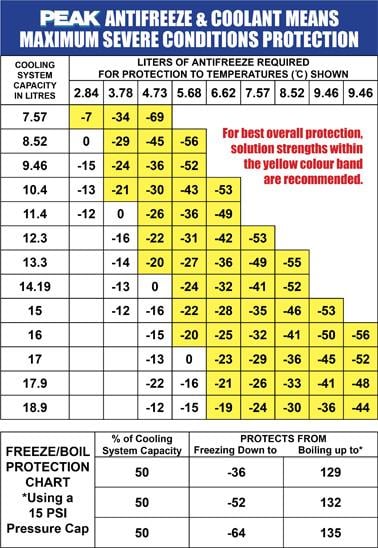

.jpg)